Difference between revisions of "Garcia-Souza 2017 MiPschool Obergurgl"
| (18 intermediate revisions by 5 users not shown) | |||
| Line 1: | Line 1: | ||
{{Abstract | {{Abstract | ||
|title=[[File:GarciaL.JPG|left|90px|Luiz Garcia-Souza]] Assessment of mitochondrial respiratory function in cryopreserved platelets. | |title=[[File:GarciaL.JPG|left|90px|Luiz Garcia-Souza]] Assessment of mitochondrial respiratory function in cryopreserved platelets. | ||
|info=[[ | |info=[[MitoEAGLE]] | ||
|authors=Garcia-Souza LF, | |authors=Garcia-Souza LF, Cizmarova B, Sumbalova Z, Menz V, Burtscher M, Gnaiger E | ||
|year=2017 | |year=2017 | ||
|event=MiPschool Obergurgl 2017 | |event=MiPschool Obergurgl 2017 | ||
|abstract=[[Image:MITOEAGLE-logo.jpg|left|100px|link=http://www.mitoglobal.org/index.php/MITOEAGLE|COST Action | |abstract=[[Image:MITOEAGLE-logo.jpg|left|100px|link=http://www.mitoglobal.org/index.php/MITOEAGLE|COST Action MitoEAGLE]] | ||
Peripheral-blood mononuclear cells (PBMC) and platelets (PLT) are potentially powerful models for the diagnosis of mitochondrial respiratory function and dysfunction, offering a minimally invasive approach in comparison to tissue biopsies. Rapid isolation of platelets and respiratory measurement without delay are required to avoid post-blood harvesting stress and cellular activation followed by metabolic alterations. Although it is well known that PLT are sensitive to temperature fluctuations, tending to activate the cells below 18 ºC [1], we considered cryopreservation as a promising strategy to preserve mitochondrial function in PLT comparable to PBMC [2]. Cryopreservation of PLT is successful for the study of inflammatory properties [3,4]. Therefore, the objective of our study was to optimize this cryopreservation protocol for the measurement of respiratory mitochondrial properties of human PLT.<br /> | Peripheral-blood mononuclear cells (PBMC) and platelets (PLT) are potentially powerful models for the diagnosis of mitochondrial respiratory function and dysfunction, offering a minimally invasive approach in comparison to tissue biopsies. Rapid isolation of platelets and respiratory measurement without delay are required to avoid post-blood harvesting stress and cellular activation followed by metabolic alterations. Although it is well known that PLT are sensitive to temperature fluctuations, tending to activate the cells below 18 ºC [1], we considered cryopreservation as a promising strategy to preserve mitochondrial function in PLT comparable to PBMC [2]. Cryopreservation of PLT is successful for the study of inflammatory properties [3,4]. Therefore, the objective of our study was to optimize this cryopreservation protocol for the measurement of respiratory mitochondrial properties of human PLT.<br /> | ||
Human blood cells were isolated in 50 ml Leucosep® tubes with 15 ml of Ficoll-Paque™. 18 ml full blood were diluted 1:1 with sterile DPBS and centrifuged at 1,000 ''g'' (first centrifugation: 10 min, room temperature, RT, acceleration 6, no brakes). The PBMC-PLT layer was collected and washed with 25 ml DPBS (120 ''g''; 10 min, RT). The supernatant was combined with 5 ml of diluted plasma obtained from the first centrifugation, and EGTA was added at 10 mM final concentration. After centrifugation at 1,000 ''g'' (10 min, RT) the cell pellet was washed with 5 ml DPBS containing 10 mM EGTA (1,000 ''g''; 5 min, RT), resuspended with 0.5 ml DPBS containing 10 mM EGTA and the PLT were counted (SYSMEX XN-350). Cells were resuspended in autologous plasma+5% DMSO for cryopreservation. Cryovials were placed in pre-cooled (4 °C) CoolCell® containers and stored at -80 °C. After one, two and four weeks storage, cells were thawed in a 37 °C water bath, diluted in 10 ml of pre-warmed 37 °C medium (DPBS+10 mM EGTA), centrifuged (1,000 ''g''; 10 min, RT) and resuspended in 0.25 ml DPBS+10 mM EGTA. Stock suspensions of cryopreserved or freshly isolated cells were titrated into the 2-ml chambers of the | Human blood cells were isolated in 50 ml Leucosep® tubes with 15 ml of Ficoll-Paque™. 18 ml full blood were diluted 1:1 with sterile DPBS and centrifuged at 1,000 ''g'' (first centrifugation: 10 min, room temperature, RT, acceleration 6, no brakes). The PBMC-PLT layer was collected and washed with 25 ml DPBS (120 ''g''; 10 min, RT). The supernatant was combined with 5 ml of diluted plasma obtained from the first centrifugation, and EGTA was added at 10 mM final concentration. After centrifugation at 1,000 ''g'' (10 min, RT) the cell pellet was washed with 5 ml DPBS containing 10 mM EGTA (1,000 ''g''; 5 min, RT), resuspended with 0.5 ml DPBS containing 10 mM EGTA and the PLT were counted (SYSMEX XN-350). Cells were resuspended in autologous plasma+5% DMSO for cryopreservation. Cryovials were placed in pre-cooled (4 °C) CoolCell® containers and stored at -80 °C. After one, two and four weeks storage, cells were thawed in a 37 °C water bath, diluted in 10 ml of pre-warmed 37 °C medium (DPBS+10 mM EGTA), centrifuged (1,000 ''g''; 10 min, RT) and resuspended in 0.25 ml DPBS+10 mM EGTA. Stock suspensions of cryopreserved or freshly isolated cells were titrated into the 2-ml chambers of the Oroboros O2k (Oroboros Instruments, Innsbruck, Austria) containing mitochondrial respiration medium MiR05 (Oroboros [[MiR05-Kit]]) or culture medium M199. An extended substrate-uncoupler-inhibitor titration (SUIT) protocol was developed using MiR05 and adding cells (ce) for measuring endogenous ROUTINE respiration (1ce), with sequential addition of pyruvate (2P; exogenous substrate), uncoupler (3U; ET capacity), glucose (4Glc; Crabtree effect), malate (5M; no stimulation expected in intact cells), rotenone, (6Rot; residual oxygen consumption, ROX, except for stimulation by PM in permeabilized cells), succinate (7S; stimulation of permeabilized cells), digitonin (8Dig; permeabilization of the plasma membrane in the entire cell population, providing a reference level of 100% nonviable cells in the respirometric cell viability test), cytochrome ''c'' (8c; test of outer mt-membrane integrity), antimycin A (9Ama; ROX), ascorbate&TMPD (10AsTm; CIV activity plus autooxidation), and azide (11Azd; chemical background to correct for autooxidation).<br /> | ||
There was no significant difference in ROUTINE respiration, ''R'', of freshly isolated PLT in M199 and MiR05. Pyruvate addition resulted in a significant increase in ''R'' in MiR05. Cold storage stress was detected as an overall decrease in mitochondrial respiration in both media, without a difference in the 1- to 4-weeks cryopreservation groups (MiR05). Flux control ratios (''FCR'') of cryopreserved and freshly isolated PLT were preserved, suggesting a global decrease in mitochondrial respiration per cell, without qualitative changes detected in our SUIT protocol. Cell viability of cryopreserved platelets decreased by 20% based on succinate respiration normalized by digitonin titration. This is consistent with an over-all loss of mitochondrial function in the non-viable cells, if permeabilization of these cells occurs in high-Ca<sup>2+</sup> medium during rewarming at 37 ºC. <br /> | There was no significant difference in ROUTINE respiration, ''R'', of freshly isolated PLT in M199 and MiR05. Pyruvate addition resulted in a significant increase in ''R'' in MiR05. Cold storage stress was detected as an overall decrease in mitochondrial respiration in both media, without a difference in the 1- to 4-weeks cryopreservation groups (MiR05). Flux control ratios (''FCR'') of cryopreserved and freshly isolated PLT were preserved, suggesting a global decrease in mitochondrial respiration per cell, without qualitative changes detected in our SUIT protocol. Cell viability of cryopreserved platelets decreased by 20% based on succinate respiration normalized by digitonin titration. This is consistent with an over-all loss of mitochondrial function in the non-viable cells, if permeabilization of these cells occurs in high-Ca<sup>2+</sup> medium during rewarming at 37 ºC. <br /> | ||
These results illustrate that cryopreservation imposes a substantial damage on PLT. Modifications, such as DMSO concentration and freezing medium cell density, will be investigated in order to improve cell viability and improve cryopreservation of mitochondrial respiratory function. | These results illustrate that cryopreservation imposes a substantial damage on PLT. Modifications, such as DMSO concentration and freezing medium cell density, will be investigated in order to improve cell viability and improve cryopreservation of mitochondrial respiratory function. | ||
|editor=[[Garcia | |editor=[[Garcia-Souza LF]], [[Kandolf G]] | ||
|mipnetlab=AT Innsbruck | |mipnetlab=AT Innsbruck Oroboros | ||
}} | }} | ||
{{Labeling | {{Labeling | ||
| Line 21: | Line 21: | ||
|organism=Human | |organism=Human | ||
|tissues=Blood cells, Platelet | |tissues=Blood cells, Platelet | ||
|preparations= | |preparations=Permeabilized cells, Intact cells | ||
|couplingstates=ROUTINE, | |couplingstates=ROUTINE, ET | ||
|pathways=S, CIV, ROX | |pathways=S, CIV, ROX | ||
|instruments=Oxygraph-2k | |instruments=Oxygraph-2k | ||
|event=C1, Oral | |event=C1, Oral | ||
|additional=PBMCs | |additional=MitoEAGLE, PBMCs | ||
}} | }} | ||
== Affiliations == | == Affiliations and support == | ||
:::: Garcia-Souza LF(1,2), | :::: Garcia-Souza LF(1,2), Cizmarova B(2,3), Sumbalova Z(2,4), Menz V(1), Burtscher M(1), Gnaiger E(2,5) | ||
::::#Inst Sport Science, Univ Innsbruck, Austria | ::::#Inst Sport Science, Univ Innsbruck, Austria | ||
::::#Daniel Swarovski Research Lab, Dept Visceral, Transplant Thoracic Surgery, Medical Univ Innsbruck, Austria | ::::#Daniel Swarovski Research Lab, Dept Visceral, Transplant Thoracic Surgery, Medical Univ Innsbruck, Austria | ||
::::#Dept Medical Clinical Biochem, Fac Medicine, Pavol Jozef Šafárik | ::::#Dept Medical Clinical Biochem, Fac Medicine, Pavol Jozef Šafárik Univ Košice, Slovakia | ||
::::#Pharmacobiochemical Lab, 3rd Dept Internal Medicine, Fac Medicine, Comenius Univ, Bratislava, Slovakia | ::::#Pharmacobiochemical Lab, 3rd Dept Internal Medicine, Fac Medicine, Comenius Univ, Bratislava, Slovakia | ||
::::# | ::::#Oroboros Instruments, Innsbruck, Austria. - [email protected] | ||
::::::Supported by [[K-Regio MitoFit]]. Contribution to European Union Framework Programme Horizon 2020 COST Action CA15203 [[MitoEAGLE]]. | |||
== Figure 1 == | == Figure 1 == | ||
[[File:Protocol Intact PLT Luiz.png|left|650px]] | [[File:Protocol Intact PLT Luiz.png|left|650px]] | ||
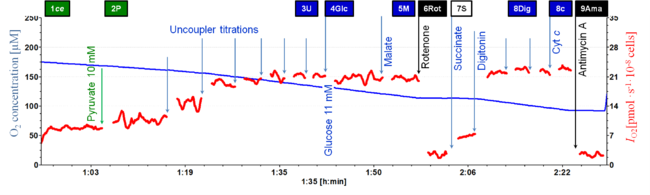
'''Figure 1.''' | '''Figure 1. Representative experiment of extended substrate-uncoupler-inhibitor titration (SUIT) protocol.''' Blue line corresponds to oxygen concentration (µM) in the chamber and red line corresponds to oxygen flow of intact platelets (pmol •s<sup>-1</sup>•10<sup>-8</sup> cells). The titration events and marks are represented as follows: ROUTINE respiration (R) is initially measured in intact cells (1ce) in mitochondrial respiration medium (MiR05-Kit); single titration of pyruvate (2P, 10 mM) to supplement MiR05 medium with oxidative substrate; uncoupler (3U*, titration of CCCP) is used for determination of ET capacity; glucose (4Glc, 11 mM) to compare Crabtree effect with culture media; malate (5M, 2 mM) stimulation is observed only in permeabilized cells; rotenone (6Rot, 0.5 mM) inhibiting Complex I (CI) in intact cells; succinate (7S, 10 mM) stimulation of mt-respiration is observed in non-viable cells only; digitonin (8Dig) is used for permeabilization of the eintire cell population, providing a reference state of succinate-stimulated ET capacity; cytochrome ''c'' (8c) titration test provides information about outer mt-membrane integrity; antimycin A (Ama, 2.5 mM) inhibiting Complex III; ascorbate 2 mM & TMPD 0.5 mM (10AsTm) stimulation of Complex IV plus chemical background; azide (11Azd, 200 mM) as an inhibitor of cytochrome c oxidase provides information about the oxygen consumption chemical background subtracted from flux in 10AsTm. | ||
== References | |||
== References == | |||
::::#Straub A, Breuer M, Wendel HP, Peter K, Dietz K, Ziemer G (2007) Critical temperature ranges of hypothermia-induced platelet activation: possible implications for cooling patients in cardiac surgery. Thromb Haemost 97:608-16. | ::::#Straub A, Breuer M, Wendel HP, Peter K, Dietz K, Ziemer G (2007) Critical temperature ranges of hypothermia-induced platelet activation: possible implications for cooling patients in cardiac surgery. Thromb Haemost 97:608-16. | ||
::::#Karabatsiakis A, Böck C, Salinas-Manrique J, Kolassa S, Calzia E, Dietrich DE, Kolassa IT (2014) Mitochondrial respiration in peripheral blood mononuclear cells correlates with depressive subsymptoms and severity of major depression. Transl Psychiatry 4:e397. | ::::#Karabatsiakis A, Böck C, Salinas-Manrique J, Kolassa S, Calzia E, Dietrich DE, Kolassa IT (2014) Mitochondrial respiration in peripheral blood mononuclear cells correlates with depressive subsymptoms and severity of major depression. Transl Psychiatry 4:e397. | ||
::::#Johnson L, Tan S, Wood B, Davis A, Marks DC (2016) Refrigeration and cryopreservation of platelets differentially affect platelet metabolism and function: a comparison with conventional platelet storage conditions. Transfusion 56:1807-18. | ::::#Johnson L, Tan S, Wood B, Davis A, Marks DC (2016) Refrigeration and cryopreservation of platelets differentially affect platelet metabolism and function: a comparison with conventional platelet storage conditions. Transfusion 56:1807-18. | ||
::::#Johnson L, Coorey CP, Marks DC (2014) The hemostatic activity of cryopreserved platelets is mediated by phosphatidylserine‐expressing platelets and platelet microparticles. Transfusion 54:1917-26. | ::::#Johnson L, Coorey CP, Marks DC (2014) The hemostatic activity of cryopreserved platelets is mediated by phosphatidylserine‐expressing platelets and platelet microparticles. Transfusion 54:1917-26. | ||
Latest revision as of 14:36, 5 July 2023
| Assessment of mitochondrial respiratory function in cryopreserved platelets. |
Link: MitoEAGLE
Garcia-Souza LF, Cizmarova B, Sumbalova Z, Menz V, Burtscher M, Gnaiger E (2017)
Event: MiPschool Obergurgl 2017
Peripheral-blood mononuclear cells (PBMC) and platelets (PLT) are potentially powerful models for the diagnosis of mitochondrial respiratory function and dysfunction, offering a minimally invasive approach in comparison to tissue biopsies. Rapid isolation of platelets and respiratory measurement without delay are required to avoid post-blood harvesting stress and cellular activation followed by metabolic alterations. Although it is well known that PLT are sensitive to temperature fluctuations, tending to activate the cells below 18 ºC [1], we considered cryopreservation as a promising strategy to preserve mitochondrial function in PLT comparable to PBMC [2]. Cryopreservation of PLT is successful for the study of inflammatory properties [3,4]. Therefore, the objective of our study was to optimize this cryopreservation protocol for the measurement of respiratory mitochondrial properties of human PLT.
Human blood cells were isolated in 50 ml Leucosep® tubes with 15 ml of Ficoll-Paque™. 18 ml full blood were diluted 1:1 with sterile DPBS and centrifuged at 1,000 g (first centrifugation: 10 min, room temperature, RT, acceleration 6, no brakes). The PBMC-PLT layer was collected and washed with 25 ml DPBS (120 g; 10 min, RT). The supernatant was combined with 5 ml of diluted plasma obtained from the first centrifugation, and EGTA was added at 10 mM final concentration. After centrifugation at 1,000 g (10 min, RT) the cell pellet was washed with 5 ml DPBS containing 10 mM EGTA (1,000 g; 5 min, RT), resuspended with 0.5 ml DPBS containing 10 mM EGTA and the PLT were counted (SYSMEX XN-350). Cells were resuspended in autologous plasma+5% DMSO for cryopreservation. Cryovials were placed in pre-cooled (4 °C) CoolCell® containers and stored at -80 °C. After one, two and four weeks storage, cells were thawed in a 37 °C water bath, diluted in 10 ml of pre-warmed 37 °C medium (DPBS+10 mM EGTA), centrifuged (1,000 g; 10 min, RT) and resuspended in 0.25 ml DPBS+10 mM EGTA. Stock suspensions of cryopreserved or freshly isolated cells were titrated into the 2-ml chambers of the Oroboros O2k (Oroboros Instruments, Innsbruck, Austria) containing mitochondrial respiration medium MiR05 (Oroboros MiR05-Kit) or culture medium M199. An extended substrate-uncoupler-inhibitor titration (SUIT) protocol was developed using MiR05 and adding cells (ce) for measuring endogenous ROUTINE respiration (1ce), with sequential addition of pyruvate (2P; exogenous substrate), uncoupler (3U; ET capacity), glucose (4Glc; Crabtree effect), malate (5M; no stimulation expected in intact cells), rotenone, (6Rot; residual oxygen consumption, ROX, except for stimulation by PM in permeabilized cells), succinate (7S; stimulation of permeabilized cells), digitonin (8Dig; permeabilization of the plasma membrane in the entire cell population, providing a reference level of 100% nonviable cells in the respirometric cell viability test), cytochrome c (8c; test of outer mt-membrane integrity), antimycin A (9Ama; ROX), ascorbate&TMPD (10AsTm; CIV activity plus autooxidation), and azide (11Azd; chemical background to correct for autooxidation).
There was no significant difference in ROUTINE respiration, R, of freshly isolated PLT in M199 and MiR05. Pyruvate addition resulted in a significant increase in R in MiR05. Cold storage stress was detected as an overall decrease in mitochondrial respiration in both media, without a difference in the 1- to 4-weeks cryopreservation groups (MiR05). Flux control ratios (FCR) of cryopreserved and freshly isolated PLT were preserved, suggesting a global decrease in mitochondrial respiration per cell, without qualitative changes detected in our SUIT protocol. Cell viability of cryopreserved platelets decreased by 20% based on succinate respiration normalized by digitonin titration. This is consistent with an over-all loss of mitochondrial function in the non-viable cells, if permeabilization of these cells occurs in high-Ca2+ medium during rewarming at 37 ºC.
These results illustrate that cryopreservation imposes a substantial damage on PLT. Modifications, such as DMSO concentration and freezing medium cell density, will be investigated in order to improve cell viability and improve cryopreservation of mitochondrial respiratory function.
• Bioblast editor: Garcia-Souza LF, Kandolf G
• O2k-Network Lab: AT Innsbruck Oroboros
Labels: MiParea: Respiration
Stress:Cryopreservation Organism: Human Tissue;cell: Blood cells, Platelet Preparation: Permeabilized cells, Intact cells
Coupling state: ROUTINE, ET
Pathway: S, CIV, ROX
HRR: Oxygraph-2k
Event: C1, Oral
MitoEAGLE, PBMCs
Affiliations and support
- Garcia-Souza LF(1,2), Cizmarova B(2,3), Sumbalova Z(2,4), Menz V(1), Burtscher M(1), Gnaiger E(2,5)
- Inst Sport Science, Univ Innsbruck, Austria
- Daniel Swarovski Research Lab, Dept Visceral, Transplant Thoracic Surgery, Medical Univ Innsbruck, Austria
- Dept Medical Clinical Biochem, Fac Medicine, Pavol Jozef Šafárik Univ Košice, Slovakia
- Pharmacobiochemical Lab, 3rd Dept Internal Medicine, Fac Medicine, Comenius Univ, Bratislava, Slovakia
- Oroboros Instruments, Innsbruck, Austria. - [email protected]
- Supported by K-Regio MitoFit. Contribution to European Union Framework Programme Horizon 2020 COST Action CA15203 MitoEAGLE.
Figure 1
Figure 1. Representative experiment of extended substrate-uncoupler-inhibitor titration (SUIT) protocol. Blue line corresponds to oxygen concentration (µM) in the chamber and red line corresponds to oxygen flow of intact platelets (pmol •s-1•10-8 cells). The titration events and marks are represented as follows: ROUTINE respiration (R) is initially measured in intact cells (1ce) in mitochondrial respiration medium (MiR05-Kit); single titration of pyruvate (2P, 10 mM) to supplement MiR05 medium with oxidative substrate; uncoupler (3U*, titration of CCCP) is used for determination of ET capacity; glucose (4Glc, 11 mM) to compare Crabtree effect with culture media; malate (5M, 2 mM) stimulation is observed only in permeabilized cells; rotenone (6Rot, 0.5 mM) inhibiting Complex I (CI) in intact cells; succinate (7S, 10 mM) stimulation of mt-respiration is observed in non-viable cells only; digitonin (8Dig) is used for permeabilization of the eintire cell population, providing a reference state of succinate-stimulated ET capacity; cytochrome c (8c) titration test provides information about outer mt-membrane integrity; antimycin A (Ama, 2.5 mM) inhibiting Complex III; ascorbate 2 mM & TMPD 0.5 mM (10AsTm) stimulation of Complex IV plus chemical background; azide (11Azd, 200 mM) as an inhibitor of cytochrome c oxidase provides information about the oxygen consumption chemical background subtracted from flux in 10AsTm.
References
- Straub A, Breuer M, Wendel HP, Peter K, Dietz K, Ziemer G (2007) Critical temperature ranges of hypothermia-induced platelet activation: possible implications for cooling patients in cardiac surgery. Thromb Haemost 97:608-16.
- Karabatsiakis A, Böck C, Salinas-Manrique J, Kolassa S, Calzia E, Dietrich DE, Kolassa IT (2014) Mitochondrial respiration in peripheral blood mononuclear cells correlates with depressive subsymptoms and severity of major depression. Transl Psychiatry 4:e397.
- Johnson L, Tan S, Wood B, Davis A, Marks DC (2016) Refrigeration and cryopreservation of platelets differentially affect platelet metabolism and function: a comparison with conventional platelet storage conditions. Transfusion 56:1807-18.
- Johnson L, Coorey CP, Marks DC (2014) The hemostatic activity of cryopreserved platelets is mediated by phosphatidylserine‐expressing platelets and platelet microparticles. Transfusion 54:1917-26.