Chinopoulos 2013 J Neurosci Res: Difference between revisions
No edit summary |
No edit summary |
||
| (7 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
{{Publication | {{Publication | ||
|title=Chinopoulos C (2013) Which way does the citric acid cycle turn during hypoxia? The critical role of α-ketoglutarate dehydrogenase | |title=Chinopoulos C (2013) Which way does the citric acid cycle turn during hypoxia? The critical role of α-ketoglutarate dehydrogenase Complex. J Neurosci Res 91:1030-43. | ||
|info=https://pubmed.ncbi.nlm.nih.gov/23378250/ | |info= [https://pubmed.ncbi.nlm.nih.gov/23378250/ PMID: 23378250 Open Access] | ||
|authors=Chinopoulos | |authors=Chinopoulos Christos | ||
|year=2013 | |year=2013 | ||
|journal=J Neurosci Res | |journal=J Neurosci Res | ||
|abstract=The citric acid cycle forms a major metabolic hub and as such it is involved in many disease states involving energetic imbalance. In spite of the fact that it is being branded as a "cycle", during hypoxia, when the electron transport chain does not oxidize reducing equivalents, segments of this metabolic pathway remain operational but exhibit opposing directionalities. This serves the purpose of harnessing high-energy phosphates through matrix substrate-level phosphorylation in the absence of oxidative phosphorylation. In this Mini-Review, these segments are appraised, pointing to the critical importance of the α-ketoglutarate dehydrogenase | |abstract=The citric acid cycle forms a major metabolic hub and as such it is involved in many disease states involving energetic imbalance. In spite of the fact that it is being branded as a "cycle", during hypoxia, when the electron transport chain does not oxidize reducing equivalents, segments of this metabolic pathway remain operational but exhibit opposing directionalities. This serves the purpose of harnessing high-energy phosphates through matrix substrate-level phosphorylation in the absence of oxidative phosphorylation. In this Mini-Review, these segments are appraised, pointing to the critical importance of the α-ketoglutarate dehydrogenase Complex dictating their directionalities. | ||
|keywords=TCA cycle | |keywords=TCA cycle | ||
<br> | |||
|editor=Cardoso LHD | |editor=Cardoso LHD | ||
|mipnetlab=HU Budapest Chinopoulos C | |mipnetlab=HU Budapest Chinopoulos C | ||
}} | }} | ||
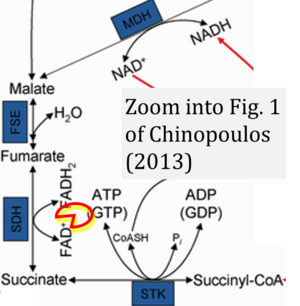
[[File:Chinopoulos 2013 J Neurosci Res CORRECTION.png|left|300px]] | |||
''Comment'': FAD is shown as FAD<sup>+</sup> - possibly by copying from NAD<sup>+</sup>. But FAD is neither charged nor a co-substrate comparable to NAD<sup>+</sup>, but it is a covalently bound prosthetic group in CII ([[Gnaiger 2023 MitoFit CII]]). | |||
{{Template:Correction FADH2 and S-pathway}} | |||
{{Labeling | {{Labeling | ||
|injuries=Hypoxia | |injuries=Hypoxia | ||
|additional=MitoFit2022rTCA | |additional=MitoFit2022rTCA | ||
}} | }} | ||
Latest revision as of 10:39, 15 October 2023
| Chinopoulos C (2013) Which way does the citric acid cycle turn during hypoxia? The critical role of α-ketoglutarate dehydrogenase Complex. J Neurosci Res 91:1030-43. |
Chinopoulos Christos (2013) J Neurosci Res
Abstract: The citric acid cycle forms a major metabolic hub and as such it is involved in many disease states involving energetic imbalance. In spite of the fact that it is being branded as a "cycle", during hypoxia, when the electron transport chain does not oxidize reducing equivalents, segments of this metabolic pathway remain operational but exhibit opposing directionalities. This serves the purpose of harnessing high-energy phosphates through matrix substrate-level phosphorylation in the absence of oxidative phosphorylation. In this Mini-Review, these segments are appraised, pointing to the critical importance of the α-ketoglutarate dehydrogenase Complex dictating their directionalities.
• Keywords: TCA cycle
• Bioblast editor: Cardoso LHD
• O2k-Network Lab: HU Budapest Chinopoulos C
Comment: FAD is shown as FAD+ - possibly by copying from NAD+. But FAD is neither charged nor a co-substrate comparable to NAD+, but it is a covalently bound prosthetic group in CII (Gnaiger 2023 MitoFit CII).
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Labels:
Stress:Hypoxia
MitoFit2022rTCA