Sharma 2021 Int J Mol Sci
| Sharma C, Kim S, Nam Y, Jung UJ, Kim SR (2021) Mitochondrial dysfunction as a driver of cognitive impairment in Alzheimer's disease. Int J Mol Sci 22:4850. https://doi.org/10.3390/ijms22094850 |
Sharma C, Kim S, Nam Y, Jung UJ, Kim SR (2021) Int J Mol Sci
Abstract: Alzheimer's disease (AD) is the most frequent cause of age-related neurodegeneration and cognitive impairment, and there are currently no broadly effective therapies. The underlying pathogenesis is complex, but a growing body of evidence implicates mitochondrial dysfunction as a common pathomechanism involved in many of the hallmark features of the AD brain, such as formation of amyloid-beta (Aβ) aggregates (amyloid plaques), neurofibrillary tangles, cholinergic system dysfunction, impaired synaptic transmission and plasticity, oxidative stress, and neuroinflammation, that lead to neurodegeneration and cognitive dysfunction. Indeed, mitochondrial dysfunction concomitant with progressive accumulation of mitochondrial Aβ is an early event in AD pathogenesis. Healthy mitochondria are critical for providing sufficient energy to maintain endogenous neuroprotective and reparative mechanisms, while disturbances in mitochondrial function, motility, fission, and fusion lead to neuronal malfunction and degeneration associated with excess free radical production and reduced intracellular calcium buffering. In addition, mitochondrial dysfunction can contribute to amyloid-β precursor protein (APP) expression and misprocessing to produce pathogenic fragments (e.g., Aβ1-40). Given this background, we present an overview of the importance of mitochondria for maintenance of neuronal function and how mitochondrial dysfunction acts as a driver of cognitive impairment in AD. Additionally, we provide a brief summary of possible treatments targeting mitochondrial dysfunction as therapeutic approaches for AD.
• Bioblast editor: Gnaiger E
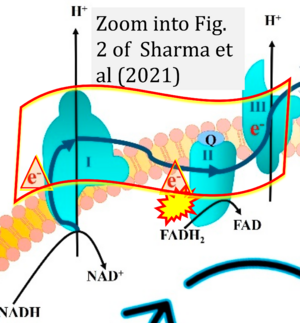
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.