Curtabbi 2022 IUBMB Life
| Curtabbi A, Enríquez JA (2022) The ins and outs of the flavin mononucleotide cofactor of respiratory complex I. IUBMB Life 74:629-44. https://doi.org/10.1002/iub.2600 |
Curtabbi A, Enríquez JA (2022) IUBMB Life
Abstract: The flavin mononucleotide (FMN) cofactor of respiratory complex I occupies a key position in the electron transport chain. Here, the electrons coming from NADH start the sequence of oxidoreduction reactions, which drives the generation of the proton-motive force necessary for ATP synthesis. The overall architecture and the general catalytic proprieties of the FMN site are mostly well established. However, several aspects regarding the complex I flavin cofactor are still unknown. For example, the flavin binding to the N-module, the NADH-oxidizing portion of complex I, lacks a molecular description. The dissociation of FMN from the enzyme is beginning to emerge as an important regulatory mechanism of complex I activity and ROS production. Finally, how mitochondria import and metabolize FMN is still uncertain. This review summarizes the current knowledge on complex I flavin cofactor and discusses the open questions for future research.
• Bioblast editor: Gnaiger E
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
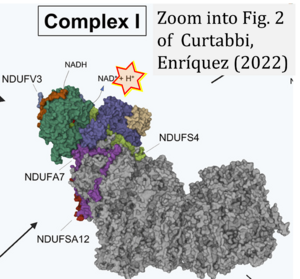
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.
Labels:
Enzyme: Complex I