Chen 2022 Int J Mol Sci
| Chen TH, Koh KY, Lin KM, Chou CK (2022) Mitochondrial dysfunction as an underlying cause of skeletal muscle disorders. Int J Mol Sci 23:12926. https://doi.org/10.3390/ijms232112926 |
Chen TH, Koh KY, Lin KM, Chou CK (2022) Int J Mol Sci
Abstract: Mitochondria are an important energy source in skeletal muscle. A main function of mitochondria is the generation of ATP for energy through oxidative phosphorylation (OXPHOS). Mitochondrial defects or abnormalities can lead to muscle disease or multisystem disease. Mitochondrial dysfunction can be caused by defective mitochondrial OXPHOS, mtDNA mutations, Ca2+ imbalances, mitochondrial-related proteins, mitochondrial chaperone proteins, and ultrastructural defects. In addition, an imbalance between mitochondrial fusion and fission, lysosomal dysfunction due to insufficient biosynthesis, and/or defects in mitophagy can result in mitochondrial damage. In this review, we explore the association between impaired mitochondrial function and skeletal muscle disorders. Furthermore, we emphasize the need for more research to determine the specific clinical benefits of mitochondrial therapy in the treatment of skeletal muscle disorders.
• Bioblast editor: Gnaiger E
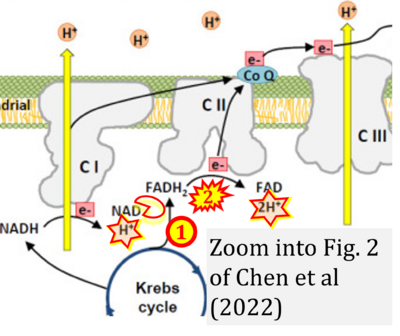
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.
Labels:
Enzyme: Complex II;succinate dehydrogenase