Speijer 2016 Biochem J
| Speijer D (2016) Being right on Q: shaping eukaryotic evolution. Biochem J 473:4103-27. |
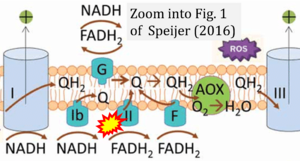
Abstract: Reactive oxygen species (ROS) formation by mitochondria is an incompletely understood eukaryotic process. I proposed a kinetic model [BioEssays (2011) 33: , 88-94] in which the ratio between electrons entering the respiratory chain via FADH2 or NADH (the F/N ratio) is a crucial determinant of ROS formation. During glucose breakdown, the ratio is low, while during fatty acid breakdown, the ratio is high (the longer the fatty acid, the higher is the ratio), leading to higher ROS levels. Thus, breakdown of (very-long-chain) fatty acids should occur without generating extra FADH2 in mitochondria. This explains peroxisome evolution. A potential ROS increase could also explain the absence of fatty acid oxidation in long-lived cells (neurons) as well as other eukaryotic adaptations, such as dynamic supercomplex formation. Effective combinations of metabolic pathways from the host and the endosymbiont (mitochondrion) allowed larger varieties of substrates (with different F/N ratios) to be oxidized, but high F/N ratios increase ROS formation. This might have led to carnitine shuttles, uncoupling proteins, and multiple antioxidant mechanisms, especially linked to fatty acid oxidation [BioEssays (2014) 36: , 634-643]. Recent data regarding peroxisome evolution and their relationships with mitochondria, ROS formation by Complex I during ischaemia/reperfusion injury, and supercomplex formation adjustment to F/N ratios strongly support the model. I will further discuss the model in the light of experimental findings regarding mitochondrial ROS formation.
• Bioblast editor: Gnaiger E
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Cited by
- Gnaiger E et al ― MitoEAGLE Task Group (2020) Mitochondrial physiology. Bioenerg Commun 2020.1. doi:10.26124/bec:2020-0001.v1.
Labels: MiParea: Respiration
Stress:Oxidative stress;RONS
Pathway: F, N, ROX
BEC 2020.1