Description
Abbreviation: NS(GM)
Reference: A: short protocol for simultaneous determination of O2 flux and mitochondrial membrane potential in mitochondrial preparations (isolated mitochondria, permeabilized cells, and tissue homogenate)-SUIT-021
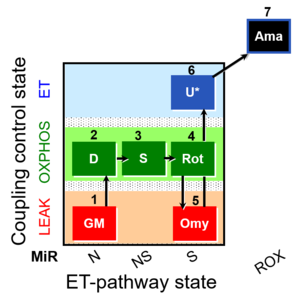
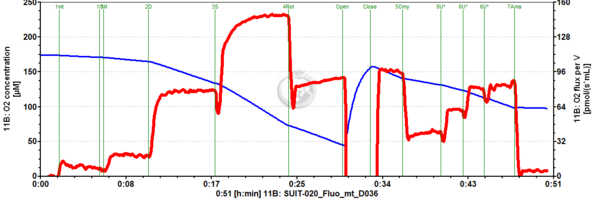
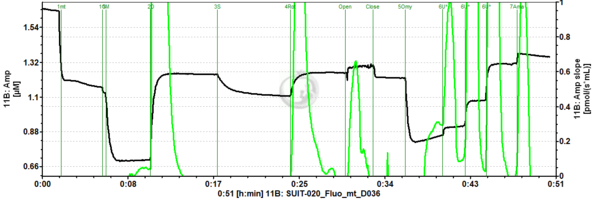
SUIT number: D036_1GM;2D;3S;4Rot;5Omy;6U;7Ama
O2k-Application: Fluo
- SUIT-category: NS(GM)
- SUIT protocol pattern: orthogonal 1GM;2D;3S;4Rot;5Omy;6U-
SUIT-021 Fluo mt D036 is a protocol to assess the additivity between N-pathway and S- pathway in the Q-junction and to investigate the N- and NS-pathway control state in mitochondrial preparations. Addition of GM (Glutamate & Malate) to the mitochondria leads to hyperpolarisation of the mt-membrane, while ADP (D) decreases the mt-membrane potential. Succinate (S) is able to further increase the mt-membrane potential. Rotenone, which is an inhibitor of Complex I, depolarises the mt-membrane. Oligomycin (Omy) is used to induce a LEAK state of respiration through inhibition of the ATP synthase. Since higher concentrations of Omy can decrease the ET state induced upon addition of uncoupler, the required concentration of Omy has to be determined. Addition of Omy results in hyperpolarisation owing to the fact that the inhibition of the ATP synthase leads to accumulation of protons in the intermembrane space. Uncoupler depolarises the mt-membrane in a concentration-dependent manner. Complex III inhibitor Antimycin (Ama) blocks the respiration. Antimycin A dissipates mt-membrane potential. For mt-membrane potential analysis and calculation, visit this page: Mitochondrial membrane potential.
Communicated by Huete-Ortega M, Komlodi T and Gnaiger E (last update 2019-07-05)
Representative traces
Steps and respiratory states
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| 1GM | GML(n) | N | CI | 1GM
|
| 2D | GMP | N | CI | 1GM;2D
|
| 3S | GMSP | NS | CI&II | 1GM;2D;3S
|
| 4Rot | SP | S | CII | 1GM;2D;3S;4Rot
|
| 5Omy | SL(Omy) | 1GM;2D;3S;4Rot;5Omy
| ||
| 6U* | SE | S | CII | 1GM;2D;3S;4Rot;5Omy;6U
|
| 7Ama | ROX | 1GM;2D;3S;4Rot;5Omy;6U;7Ama
|
- Bioblast links: SUIT protocols - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- Coupling control
- Pathway control
- Main fuel substrates
- » Glutamate, G
- » Glycerophosphate, Gp
- » Malate, M
- » Octanoylcarnitine, Oct
- » Pyruvate, P
- » Succinate, S
- Main fuel substrates
- Glossary
Strengths and limitations
- Before performing this protocol, calibration with the fluorescence dye needs to be done. More information on our USB: Instrumental Protocols/Fluo calibration.
- It is recommended to run a chemical background assay without any sample to test the effect of the chemicals on the fluorescence signal. More information on our USB.
- NS-OXPHOS capacity provides a physiologically relevant estimate of maximum mitochondrial respiratory capacity.
- Comparison of GM- with PM-capacity yields important information on N-pathway respiratory control upstream of CI.
- A succinate concentration of >10 mM may be required for saturating SE capacity.
- Nigericin as a H+/K+ antiporter can be used to dissipate transmembrane pH gradient, which results in increased mt-membrane potential in the LEAK state.
- + You obtain information in a single protocol about the NS-, S and N- pathway control state.
- - Many fluorescence dyes (such as Safranin, TMRM etc) can inhibit components of the ET system, most commonly affecting NADH-linked respiration. Therefore, a control run of the protocol should be done in the absence of the fluorescence dye. The following protocol can be used: SUIT-021 O2 mt D035.
- - To test the effect of the fluorescence dye on the respiration, in SUIT-020_O2_mt_D032 and SUIT-020_O2_mt_D035 the dye can be added after cytochrome c.
- - CIV activity cannot be determined and Cytochrome c test cannot be performed together with the fluorescence dyes.
- - Oligomycin concentration has to be determined. Higher concentrations of Omy may inhibit the ET state.
- - Careful washing is required after the experiment to avoid carry-over of the inhibitors and uncoupler.
Compare SUIT protocols
- SUIT-004;SUIT-004 O2 pfi D010: provides information about the NS(PM) pathway in the ET state without the contribution of G.
- SUIT-008;SUIT-008 O2 pfi D014 and SUIT-008 O2 pce D025: provide information about the NS(PGM) pathway in the OXPHOS and ET-states, but without the addition of Omy.
- SUIT-011: provides information about the NS(GM) pathway in the OXPHOS and ET-states, but without the addtion of Omy.
- SUIT-012: provides information about the N(PGM) pathway in the OXPHOS and ET-states without the contribution of S-pathway and without the addition of Omy.
- SUIT-014: a protocol similar to SUIT-021 O2 mt D035, but with the addition of P in the OXPHOS state before S. Omy is not added in the protocol.
Chemicals and syringes
| Step | Chemical(s) and link(s) | Comments |
|---|---|---|
| Saf | Safranin (Saf) |
or
| Step | Chemical(s) and link(s) | Comments |
|---|---|---|
| TMRM | Tetramethylrhodamine methyl ester (TMRM) |
or
| Step | Chemical(s) and link(s) | Comments |
|---|---|---|
| Rh123 | Rhodamine 123 |
| Step | Chemical(s) and link(s) | Comments |
|---|---|---|
| 1GM | Glutamate (G) and Malate (M) | |
| 2D | ADP (D) | |
| 3S | Succinate (S) | |
| 4Rot | Rotenone (Rot) | |
| 5Omy | Oligomycin (Omy) | |
| 6U | Carbonyl cyanide m-chlorophenyl hydrazone, CCCP (U) | Can be substituted for other uncoupler |
| 7Ama | Antimycin A (Ama) |
- Suggested stock concentrations are shown in the specific DL-Protocol.
References
| Year | Reference | Organism | Tissue;cell | |
|---|---|---|---|---|
| MiPNet20.13 Safranin mt-membranepotential | 2019-06-24 | O2k-FluoRespirometry: HRR and simultaneous determination of mt-membrane potential with safranin or TMRM. | Mouse | Nervous system |
| Krumschnabel 2014 Methods Enzymol | 2014 | Krumschnabel G, Eigentler A, Fasching M, Gnaiger E (2014) Use of safranin for the assessment of mitochondrial membrane potential by high-resolution respirometry and fluorometry. https://doi.org/10.1016/B978-0-12-416618-9.00009-1 | Mouse | Nervous system |
MitoPedia concepts: SUIT protocol, SUIT A, Find
MitoPedia methods:
Fluorometry