Penjweini 2020 Redox Biol
| Penjweini R, Roarke B, Alspaugh G, Gevorgyan A, Andreoni A, Pasut A, Sackett DL, Knutson JR (2020) Single cell-based fluorescence lifetime imaging of intracellular oxygenation and metabolism. Redox Biol 34:101549. https://doi.org/10.1016/j.redox.2020.101549 |
Penjweini R, Roarke B, Alspaugh G, Gevorgyan A, Andreoni A, Pasut A, Sackett DL, Knutson JR (2020) Redox Biol
Abstract: Oxidation-reduction chemistry is fundamental to the metabolism of all living organisms, and hence quantifying the principal redox players is important for a comprehensive understanding of cell metabolism in normal and pathological states. In mammalian cells, this is accomplished by measuring oxygen partial pressure (pO2) in parallel with free and enzyme-bound reduced nicotinamide adenine dinucleotide (phosphate) [H] (NAD(P)H) and flavin adenine dinucleotide (FAD, a proxy for NAD+). Previous optical methods for these measurements had accompanying problems of cytotoxicity, slow speed, population averaging, and inability to measure all redox parameters simultaneously. Herein we present a Förster resonance energy transfer (FRET)-based oxygen sensor, Myoglobin-mCherry, compatible with fluorescence lifetime imaging (FLIM)-based measurement of nicotinamide coenzyme state. This offers a contemporaneous reading of metabolic activity through real-time, non-invasive, cell-by-cell intracellular pO2 and coenzyme status monitoring in living cells. Additionally, this method reveals intracellular spatial heterogeneity and cell-to-cell variation in oxygenation and coenzyme states.
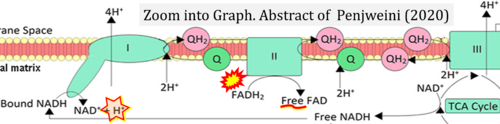
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
- Labelling FAD as 'Free FAD' is incorrect, since the prosthetic group FAD/FADH2 remains covalently bound to the subunit SDHA of CII in the catalytic cycle.
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.
Labels:
Stress:Hypoxia