Liang 2019 Front Microbiol
| Liang J, Huang H, Wang S (2019) Distribution, evolution, catalytic mechanism, and physiological functions of the flavin-based electron-bifurcating NADH-dependent reduced ferredoxin: NADP+ oxidoreductase. Front Microbiol 10:373. https://doi.org/10.3389/fmicb.2019.00373 |
Liang J, Huang H, Wang S (2019) Front Microbiol
Abstract: NADH-dependent reduced ferredoxin:NADP+ oxidoreductase (Nfn) is an electron-bifurcating enzyme first discovered in the strict anaerobes Clostridium kluyveri and Moorella thermoacetica. In vivo, Nfn catalyzes the endergonic reduction of NADP+ with NADH coupled to the exergonic reduction of NADP+ with reduced ferredoxin. Most Nfn homologs consist of two subunits, although in certain species Nfn homologs are fused. In contrast to other electron-bifurcating enzymes, Nfn possess a simpler structure. Therefore, Nfn becomes a perfect model to determine the mechanism of flavin-based electron bifurcation, which is a novel energy coupling mode distributed among anaerobic bacteria and archaea. The crystal structures of Nfn from Thermotoga maritima and Pyrococcus furiosus are known, and studies have shown that the FAD molecule of the NfnB (b-FAD) is the site of electron bifurcation, and other cofactors, including a [2Fe2S] cluster, two [4Fe4S] clusters, and the FAD molecule on the NfnA subunit, contribute to electron transfer. Further, the short-lived anionic flavin semiquinone (ASQ) state of b-FAD is essential for electron bifurcation. Nfn homologs are widely distributed among microbes, including bacteria, archaea, and probably eukaryotes, most of which are anaerobes despite that certain species are facultative microbes and even aerobes. Moreover, potential evidence shows that lateral gene transfer may occur in the evolution of this enzyme. Nfn homologs present four different structural patterns, including the well-characterized NfnAB and three different kinds of fused Nfn homologs whose detailed properties have not been characterized. These findings indicate that gene fusion/fission and gene rearrangement may contribute to the evolution of this enzyme. Under physiological conditions, Nfn catalyzes the reduction of NADP+ with NADH and reduced ferredoxin, which is then used in certain NADPH-dependent reactions. Deletion of nfn in several microbes causes low growth and redox unbalance and may influence the distribution of fermentation products. It's also noteworthy that different Nfn homologs perform different functions according to its circumstance. Physiological functions of Nfn indicate that it can be a potential tool in the metabolic engineering of industrial microorganisms, which can regulate the redox potential in vivo.
• Bioblast editor: Gnaiger E
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
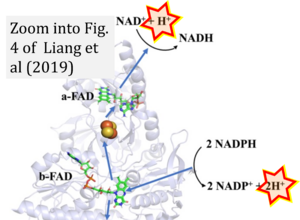
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.