Gasmi 2021 Arch Toxicol
| Gasmi A, Peana M, Arshad M, Butnariu M, Menzel A, Bjørklund G (2021) Krebs cycle: activators, inhibitors and their roles in the modulation of carcinogenesis. Arch Toxicol 95:1161-78. https://doi.org/10.1007/s00204-021-02974-9 |
Gasmi A, Peana M, Arshad M, Butnariu M, Menzel A, Bjoerklund G (2021) Arch Toxicol
Abstract: A fundamental metabolic feature of cancerous tissues is high glucose consumption. The rate of glucose consumption in a cancer cell can be 10-15 times higher than in normal cells. Isolation and cultivation of tumor cells in vitro highlight properties that are associated with intensive glucose utilization, the presence of minimal oxidative metabolism, an increase in lactate concentrations in the culture medium and a reduced rate of oxygen consumption. Although glycolysis is suggested as a general feature of malignant cells and recently identified as a possible contributing factor to tumor progression, several studies highlight distinct metabolic characteristics in some tumors, including a relative decrease in avidity compared to glucose and/or a glutamine dependency of lactate and even proliferative tumor cells. The aim of this review is to determine the particularities in the energy metabolism of cancer cells, focusing on the main nutritional substrates, such as glucose and glutamine, evaluating lactate dehydrogenase as a potential marker of malignancy and estimating activators and inhibitors in cancer treatment.
• Bioblast editor: Gnaiger E
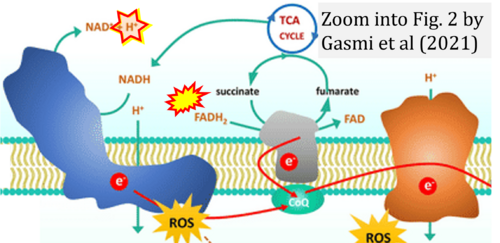
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.
Labels: MiParea: Pharmacology;toxicology
Pathology: Cancer
Pathway: S