Garlid AO 2012 Abstract Bioblast
| Garlid AO, Garlid KD (2012) Na,K-ATPase signaling to mitochondria. Mitochondr Physiol Network 17.12. |
Link: MiPNet17.12 Bioblast 2012 - Open Access
Event: Bioblast 2012
Binding of cardiac glycosides to the Na,K-ATPase has two effects: increased contractility (inotropy) and cardioprotection against ischemia-reperfusion injury [1]. Cardioprotection is mediated by caveolar vesicles (signalosomes) that bud off the plasma membrane, move to mitochondria, and use a terminal kinase to phosphorylate an outer membrane protein. This activates inner membrane PKCεs, causing opening of the mitochondrial ATP-sensitive K+ channel (mtKATP) and inhibition of the mitochondrial permeability transition (mtPT). These events are assayed in vitro using purified signalosomes from treated hearts and mitochondria from untreated hearts [2].
The terminal kinase of GPCR signalosomes is PKG [2]; however PKG does not mediate cardioprotection by ouabain, so we set out to determine the terminal kinases used by the ouabain signalosome. Rat hearts perfused with ouabain yielded a signalosome fraction that was caveolar in nature and enriched with caveolins 1 and 3, Src, PKCε and the α-1-subunit of the Na+,K+-ATPase. Electron microscopy of purified signalosomes revealed vesicles approximately 140 nM in diameter that were found by immunogold labeling to be decorated with caveolin-3. Ouabain signalosomes from heart opened mtKATP in mitochondria isolated from untreated hearts and liver. The terminal kinases of the ouabain signalosome are Src and PKCε, which together phosphorylated an endogenous outer membrane p38MAPK. We conclude (1) that ouabain cardioprotection utilizes the signalosome mechanism; (2) that the terminal kinases acting on mitochondria are Src and PKCε, (3) that this is a general mechanism of cell signaling, given that signalosomes from rat heart open mtKATP in rat liver mitochondria. (4) that ouabain cardioprotection acts via a mitochondrial p38 MAPK.
Digitalis has been used in the treatment of heart failure since 1785. It was thought for many years that its efficacy was due to its positive inotropic effect. This may not be the case. Cardioprotection in both rat and rabbit is seen with concentrations of ouabain that have no inotropic effect.
- Pasdois P, Beauvoit B, Costa AD, Vinassa B, Tariosse L, Bonoron-Adèle S, Garlid KD, Dos Santos P (2007) Sarcoplasmic ATP-sensitive potassium channel blocker HMR1098 protects the ischemic heart: implication of calcium, Complex I, reactive oxygen species and mitochondrial ATPsensitive potassium channel. J Mol Cell Cardiol 42: 631-642.
- Garlid KD, Costa AD, Quinlan CL, Pierre SV, Dos Santos P (2009) Cardioprotective signaling to mitochondria. J Mol Cell Cardiol 46: 858-866. Open Access
• Keywords: Na/K ATPase, Signaling, Ouabain, Cardioprotection, p38 MAPK, Digitalis
Labels:
Stress:Ischemia-reperfusion Organism: Rat Tissue;cell: Heart Preparation: Intact organ, Isolated mitochondria
Affiliations and author contributions
Anders O Garlid, Keith D Garlid
Department of Biology, Portland State University, Portland, Oregon, USA; Email: [email protected]
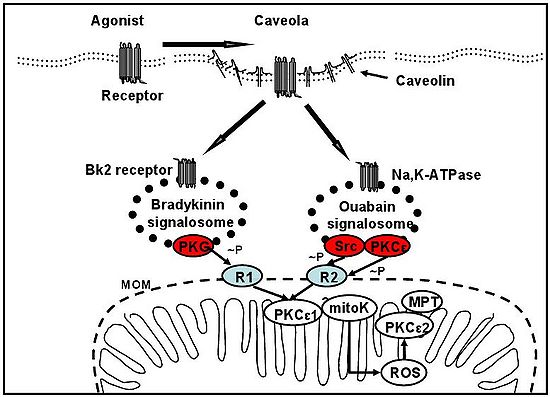
Figure 1
Signaling to mitochondria via signalosome. Upon binding to their respective receptors, bradykinin and ouabain trigger formation of caveolar signaling platforms that are encapsulated in signalosomes. Signalosomes bind to and phosphorylate mitochondrial outer membrane (mtOM; MOM) receptors via specific terminal kinases. This causes the signal to be transmitted across the mtOM and intermembrane space, to PKCε1 on the mitochondrial inner membrane and trigger the intramitochondrial signaling pathway, leading to activation of PKCε2 and inhibition of mtPT. The terminal kinase of the bradykinin signalosome is PKG, which phosphorylates a mtOM receptor (R1) of unknown identity. The terminal kinases of the ouabain signalosome are Src and PKCe acting in concert to phosphorylate a mtOM p38MAPK.