Duchen 2012 Abstract Bioblast
| Blacker TS, Gale JE, Ziegler M, Bain AJ, Szabadkai G, Duchen MR (2012) Separation of NADPH and NADH fluorescence emission in live cells using fluorescence lifetime imaging microscopy. Mitochondr Physiol Network 17.12. |
Link: MiPNet17.12 Bioblast 2012 - Open Access
Blacker TS, Gale JE, Ziegler M, Bain AJ, Szabadkai G, Duchen MR (2012)
Event: Bioblast 2012
Measurements of fluorescence intensity of endogenous NADH and NADPH have been used to monitor cellular metabolic state for ~50 years [1]. As NADH and NADPH are fluorescent and NAD+ and NADP+ are not, the fluorescence signal gives a unique measure of the redox state of the NADH:NADPH pool, reflecting the balance of substrate supply and respiratory rate at the level of the single cell. An additional dimension of information is available through measurements of fluorescence lifetime (the mean dwell time of an electron in an excited state), as lifetime measurements are exquisitely sensitive to the microenvironment of a fluorophore. Thus, the fluorescence lifetime of NADH in solution (~0.4ns) increases more than 5-fold when NADH is enzyme bound, with a precise lifetime governed by the enzyme to which the cofactor is bound (giving a mean lifetime of ~2.7ns). It is also apparent that lifetimes vary depending on cellular metabolic state, but the biochemical basis for those variations in lifetime has not been clarified (e.g.[2]).
We have used fluorescence lifetime imaging (FLIM) to explore changes in NADH and NADPH lifetimes in cells in culture under a variety of carefully controlled metabolic conditions. Lifetimes were measured using a Becker and Hickl system coupled to a Zeiss META NLO microscope using a Coherent Chameleon pulsed laser tuned to 700nm, with emission measured between 420-480nm. We have found that changes in relative glycolytic or oxidative metabolism do not change lifetimes, but do change the relative weighting of the lifetimes that arise from free and enzyme bound NADH. Further, we found that enzyme bound NADH and NADPH, two species which are functionally distinct but spectrally identical, show distinct lifetimes, allowing separation of NADH and NADPH pools in cells and tissues. Thus, in HEK cells stably overexpressing NAD kinase (NADK), in which NADPH levels are elevated about 15 fold [3], the fluorescence lifetime of free NAD(P)H was unchanged at ~0.4ns, but the fluorescence lifetime arising from enzyme-bound NADPH was greatly increased to 3.9 ± 0.4 ns compared to 3.0 ±0.2 ns measured in HEK cells in which NADK was stably knocked-down.
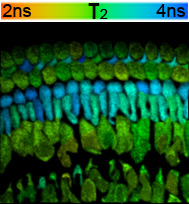
FLIM studies in complex preparations (brain slices, cochlear explant cultures, renal slices etc) revealed individual cell types enriched in NADPH. Thus, FLIM imaging of cochlear explant cultures from postnatal day 2-3 rats revealed a subset of supporting cells showing an equivalent long lifetime (mean 3.6ns), suggesting high levels of NADPH in these cells (Fig 1). The long lifetime signal was reduced by exposure to the drug Epigallocatechin-3-gallate (EGCG) which displaces NADPH from its binding sites, supporting this conclusion.
These data suggest that FLIM provides a unique and novel approach to separate NADH and NADPH signals, at last providing an approach to address specific changes in NADPH and NADH and their contributions to physiology in living cells and tissues.
• Keywords: NADH, Redox, Fluorescence lifetime imaging, HEK cells, Cochlear explant cultures
• O2k-Network Lab: UK_London_Duchen MR
Labels:
Organism: Human, Rat
Tissue;cell: Nervous system, HEK
Affiliations and author contributions
Tom S Blacker (1,2), Jonathan E Gale (1,3), Matthias Ziegler (4), Angus J Bain (2), Gyorgy Szabadkai (1), Michael R Duchen (1)
(1) Department of Cell & Developmental Biology, University College London, UK Email: [email protected]
(2) Department of Physics and Astronomy, University College London, UK
(3) Ear Institute, University College London, UK
(4) Department of Molecular Biology, University of Bergen, Norway
From the UCL Consortium for Mitochondrial Research
Acknowledgements: This work is supported by a PhD studentship to TB funded by the EPSRC and CoMPLEX at UCL.
Figure 1
The image shows the distribution of long (enzyme bound) lifetimes (tau 2) of NADH; NADPH acquired from a rat cochlear explant culture. Excitation was at 700nm, emission acquired between 420-480nm. Lifetimes are colour coded between 2-4ns graded from red to blue, as indicated. Note the layer of cells with lifetimes close to 4ns, in contrast to the majority in which the mean long lifetime is close to 3ns.