Crisostomo 2017 MiP2017
| Glycerol metabolism and mitochondrial function in human Sertoli cells - a preliminary study. |
Link: MiP2017
Crisostomo L, Jarak I, Carvalho RA, Sousa M, Barros A, Alves MG, Oliveira PF (2017)
Event: MiP2017
Metabolic diseases have rocketed worldwide during the last decades. During the same period, fertility rates have withered proportionally. Poor food habits and sedentarism are usually regarded as a shared cause of both tendencies. Indeed, sexual function is sensitive to energy balance, although the biochemical link between metabolic disease and reproductive (dys)function is not well-understood [1]. Glycerol is a by-product of dietary fats, namely from triacylglycerols. This small molecule poses virtually no cytotoxicity, but it was found that injecting a solution with high glycerol concentration in tests results in arrestment of spermatogenesis [2]. Besides, glycerol is a metabolite that links the metabolic pathways for carbohydrates and lipids, acting as a metabolic rerouter [3]. Sertoli cells present the biochemical machinery to import and metabolize glycerol [4], thus it might play a crucial role on their homeostasis and spermatogenesis.
In this study, we investigated the potential role of glycerol on human Sertoli cells' (hSC) metabolism. Primary hSC cultures were established after hSC isolation from 6 subjects with conserved spermatogenesis subjected to testicular biopsy for spermatozoa retrieval, due to anajeculation or previous vasectomy. Once cultures reached 70%-80% confluence, we divided the cells in 4 groups, and incubated them for 24h. One group was then grown in Sertoli cell culture medium, whilst the others had it replaced by glycerol supplemented medium at different concentration (infraphysiologic – 0.1 µM, physiologic – 10µM and superphysiologic – 1000 µM). After this period, cells were tested for cytotoxicity (LDH assay), viability (MTT assay), mitochondrial functionality (JC1 assay) and protein expression of mitochondrial complexes (Western Blot). We also collected culture medium before adding to cultures, and extracellular medium after the 24h-period, for metabolite quantification (1H-NMR), in order to establish the metabolic profile (excretion or production) of the cells.
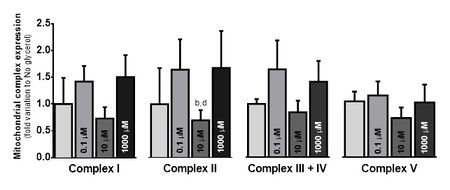
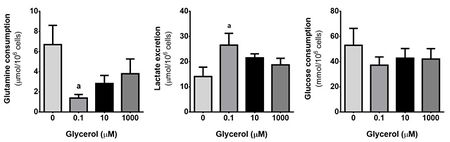
Our results showed a significant cytoprotective effect of glycerol at 0.1 µM (p < 0.05), but no changes were observed in cell viability between groups. We did not find any difference in mitochondrial membrane potential between the different groups, though Complex II protein expression was significantly lower at 10 µM than at 1000 µM (p < 0.05) (Fig. 1). Lactate was significantly more excreted in the 0.1 µM glycerol cells, while glutamine was significantly less consumed in the same group (p < 0.05). Interestingly, glucose consumption showed no differences between groups (Fig. 2).
Our results show that glutaminolysis, an important metabolic pathway in hSCs for lactate production, is repressed in cells maintained in glycerol-depleted medium. However, lactate excretion is enhanced in this case, without any contribution from glucose nor cell death, since there was no cytotoxicity in these cells. Therefore, lactate should be obtained from an alternative source. Complex II overexpression at higher glycerol concentration may indicate a promotion of succinate pathway, although its source is not clear. Overall, our data suggests that glycerol has a concentration-dependent effect on mitochondrial function and metabolic behaviour of hSCs. Since hSCs are responsible for the nutritional support of spermatogenesis, glycerol may be a novel modulator of this process that determines male fertility.
• Bioblast editor: Kandolf G
Labels: MiParea: Exercise physiology;nutrition;life style, Pharmacology;toxicology
Organism: Human
Tissue;cell: Genital
Enzyme: Complex II;succinate dehydrogenase
Figures
Figure 1: Electron Transfer Chain complex protein expression (fold increase to “No glycerol” group. Complex II was the only one which presented significant differences between groups (p < 0.05). A superphysiological glycerol concentration (1000 µM)induced a Complex II protein expression significantly higher than a physiological concentration (10 µM). This data may indicate that high glycerol availability overcharges Succinate dehydrogenase. An increase in Complex II activity, which is poorly protected against electron leak, may lead to increased oxidative stress in mitochondria. a) relative to No glycerol; b) relative to 0.1 µM glycerol; c) relative to 10 µM glycerol; d) relative to 1000 µM glycerol
Figure 2: Metabolic profile of hSC primary cultures after 24h in glycerol supplemented medium. Significance was considered when p < 0.05. When glycerol was present at lower than physiological concentration (0.1 µM), glutamine consumption decreased but, interestingly, lactate production was promoted. Even more interesting, glucose consumption was not affected. These data indicates a suppression of glutaminolysis, however, it is not clear which metabolic pathway is compensating it to foster lactate excretion. It is possible that lipid metabolism might be favoured in this condition. a) relative to No glycerol; b) relative to 0.1 µM glycerol; c) relative to 10 µM glycerol; d) relative to 1000 µM glycerol
Affiliations
- Crisóstomo L(1,2,3), Jarak I(1,4), Carvalho RA(4), Sousa M(1,4), Barros A(2,3,5), Alves MG(1), Oliveira PF(1,2,3)
- Dept Microscopy, Lab Cell Biology, and Unit Multidisciplinary Research Biomedicine (UMIB), Inst Biomedical Sciences Abel Salazar (ICBAS), Univ Porto, Portugal
- Dept Genetics, Fac Medicine, Porto, Portugal
- i3S – Inst Investigação Inovação em Saúde, Univ Porto, Portugal
- Dept Life Sciences, Fac Sciences Technol, Univ Coimbra, Portugal
- Centre Reproductive Genetics Prof. Alberto Barros, Porto, Portugal. – [email protected]
References
- Oliveira PF, Sousa M, Silva BM, Monteiro MP, Alves MG (2017) Obesity, energy balance and spermatogenesis. Reproduction 153:173-85.
- Wiebe JP, Kowalik A, Gallardi RL, Egeler O, Clubb BH (2000) Glycerol disrupts tight junction-associated actin microfilaments, occludin, and microtubules in Sertoli cells. J Andrology 21:625-35.
- Crisóstomo L, Alves MG, Calamita G, Sousa M, Oliveira PF (2017) Glycerol and testicular activity: the good, the bad and the ugly. Molecular human reproduction 23.
- Jesus TT, Bernardino RL, Martins AD, Sá R, Sousa M, Alves MG, Oliveira PF (2014) Aquaporin-9 is expressed in rat Sertoli cells and interacts with the cystic fibrosis transmembrane conductance regulator. IUBMB Life 66:639-44.