Chowdhury 2018 Oxid Med Cell Longev
| Roy Chowdhury S, Banerji V (2018) Targeting mitochondrial bioenergetics as a therapeutic strategy for chronic lymphocytic leukemia. Oxid Med Cell Longev 2018:2426712. https://doi.org/10.1155/2018/2426712 |
Roy Chowdhury S, Banerji V (2018) Oxid Med Cell Longev
Abstract: Altered cellular metabolism is considered a hallmark of cancer and is fast becoming an avenue for therapeutic intervention. Mitochondria have recently been viewed as an important cellular compartment that fuels the metabolic demands of cancer cells. Mitochondria are the major source of ATP and metabolites necessary to fulfill the bioenergetics and biosynthetic demands of cancer cells. Furthermore, mitochondria are central to cell death and the main source for generation of reactive oxygen species (ROS). Overall, the growing evidence now suggests that mitochondrial bioenergetics, biogenesis, ROS production, and adaptation to intrinsic oxidative stress are elevated in chronic lymphocytic leukemia (CLL). Hence, recent studies have shown that mitochondrial metabolism could be targeted for cancer therapy. This review focuses the recent advancements in targeting mitochondrial metabolism for the treatment of CLL.
• Bioblast editor: Gnaiger E
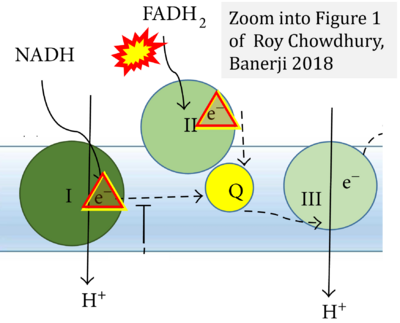
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.
Labels: MiParea: Respiration, Pharmacology;toxicology
Pathology: Cancer