Description
Abbreviation: O2 dependence-H2O2
Reference: A: A protocol for oxygen dependence of O2 flux and H2O2 flux in isolated mitochondria,tissue homogenate or permeabilized cells
- SUIT protocol pattern: 1GMS;2D;-
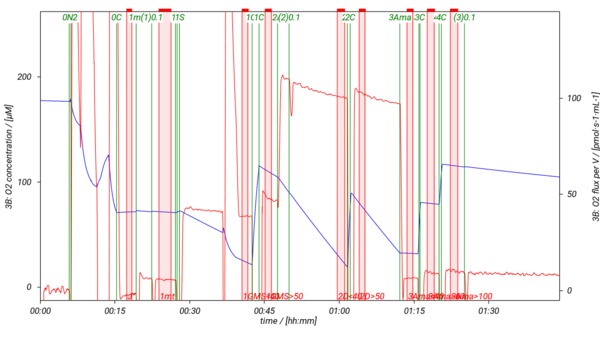
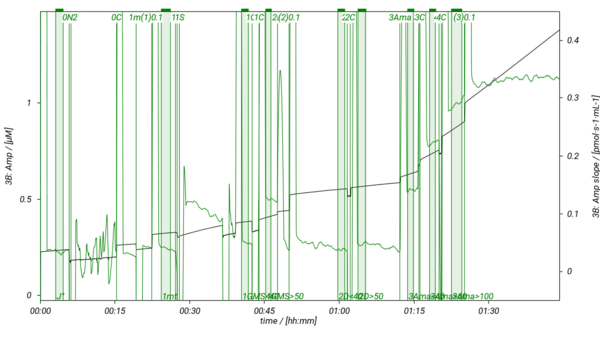
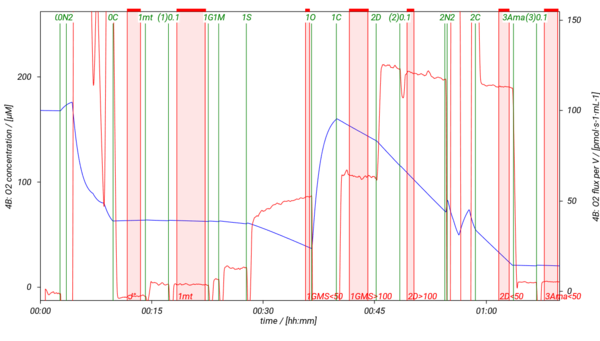
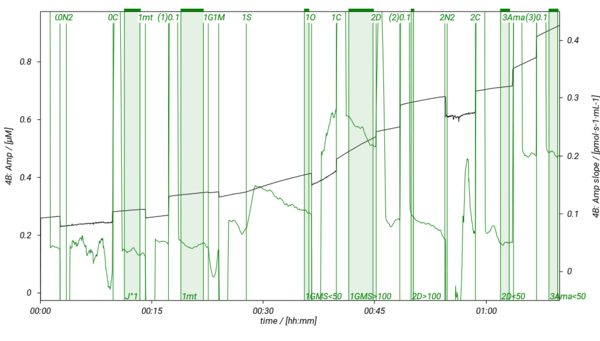
SUIT-018 is a short protocol to study simultaneously the oxygen dependence of O2 flux and H2O2 flux in isolated mitochondria, tissue homogenate (except of liver homogenate) or permeabilized cells. In our experiments H2O2 flux shows linear dependence on oxygen concentration in LEAK, OXPHOS, ET states and in ROX state measured in MiR05-Kit, therefore, we recommend varying the oxygen level in the O2k-Chamber several times during the experiment. It is advisable to detect H2O2 flux in the same state either at high oxygen concentration (tissue hyperoxia; [O2]> ~100-110 µM) and at low oxyen concentration (tissue normoxia; [O2]< ~70-80 µM), or keep always the oxygen level in tisse normoxia. In this SUIT protocol several DLPs are listed to evaluate H2O2 flux in the NS-pathway in the LEAK and OXPHOS states. To decrease or increse the oxygen concentration in the O2k-Chamber, see Setting the oxygen concentration.
Communicated by Komlodi T, Gnaiger E (last update 2020-06-09)
Specific SUIT protocols
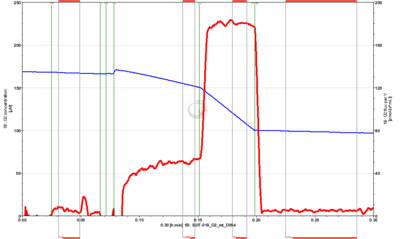
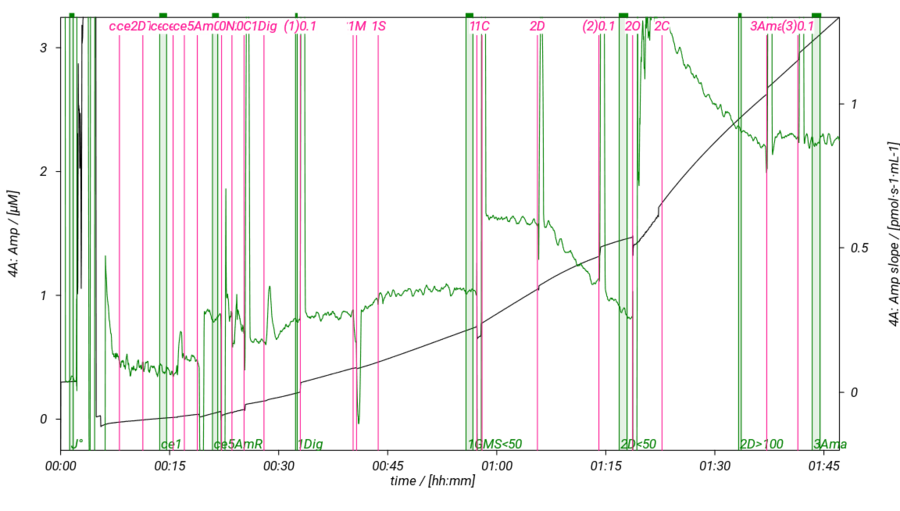
SUIT-018 AmR mt D031
- SUIT-018 AmR mt D031 simultaneous determination of O2 and H2O2 flux in isolated mitochondria, tissue homogenate and permeabilized cells at low oxygen concentration (tissue normoxia).
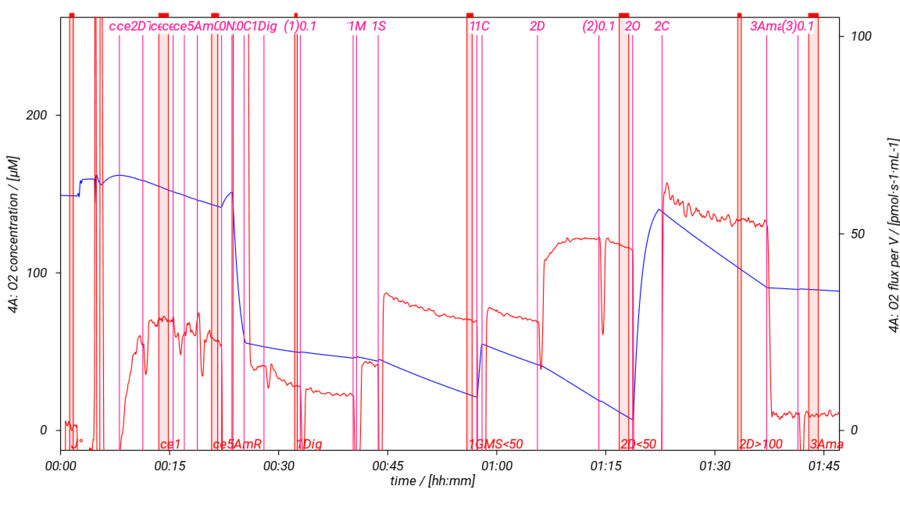
SUIT-018 AmR mt D040
- SUIT-018 AmR mt D040 simultaneous determination of O2 and H2O2 flux in isolated mitochondria, tissue homogenate and permeabilized cells at various oxygen levels.
SUIT-018 AmR mt D041
- SUIT-018 AmR mt D041 simultaneous determination of O2 and H2O2 flux in isolated mitochondria, tissue homogenate and permeabilized cells changing the oxygen concentration in the same protocol.
SUIT-018 O2 mt D054
- SUIT-018 O2 mt D054 to measure O2 flux in isolated mitochondria, tissue homogenate and permeabilized cells as a control to test the effect of the fluorescence dye on the respiration.
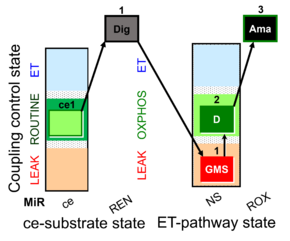
SUIT-018 AmR ce-pce D068
- SUIT-018 AmR ce-pce D068 simultaneous determination of O2 and H2O2 flux in permeabilized cells changing the oxygen concentration in the same protocol with testing the effect of the fluorescence dye on the ROUTINE-respiration.
Steps and respiratory states
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| 0DTPA |
| |||
| 0SOD |
| |||
| 0HRP |
| |||
| 0AmR |
|
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
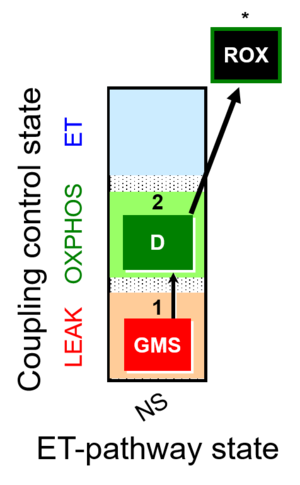
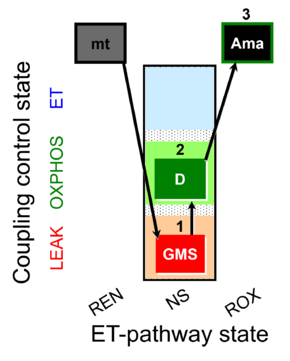
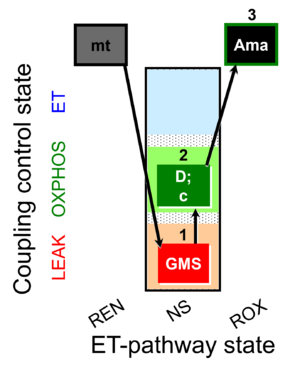
| 1GMS | GMSL(n) | NS | CI&CII | 1GMS
|
| 2D | GMSP | NS | CI&CII | 1GMS;2D
|
| 3Ama | ROX | 1GMS;2D;3Ama
|
Strengths and limitations
- Many fluorescence dyes can inhibit components of the ET system and thus, affecting the respiration. Therefore, a control run of the protocol should be done in the absence of the fluorescence dye.
- + Short duration of the experiment.
- + Simple protocol to evaluate the oxygen dependence of H2O2 production in LEAK and OXPHOS state.
- - Measurements with Amplex UltraRed assay cannot be carried out with liver homogenate.
- - Reoxygenation and nitrogen injection could lead to bubble formation in the O2k-Chamber.
- - CIV activity and cytochrome c test cannot be performed together with the fluorescence.
Compare SUIT protocols
- SUIT-006 AmR mt D048 to investigate the dependence of H2O2 flux on the mt-membrane potential on the N-control state on isolated mitochondria and tissue homogenate.
- SUIT-009 to investigate H2O2 production driven by the reverse electron transfer (RET) on isolated mitochondria, tissue homogenate and permeabilized cells.
References
| Year | Reference | Organism | Tissue;cell | |
|---|---|---|---|---|
| MiPNet24.10 H2O2 flux analysis | 2021-10-22 | Hydrogen peroxide flux analysis using Amplex UltraRed assay in MiR05-Kit with DatLab 7.4 | ||
| Komlodi 2021 BEC AmR-O2 | 2021 | Komlódi T, Sobotka O, Gnaiger E (2021) Facts and artefacts on the oxygen dependence of hydrogen peroxide production using Amplex UltraRed. Bioenerg Commun 2021.4. https://doi.org/10.26124/bec:2021-0004 | Saccharomyces cerevisiae | Other cell lines |
| MiPNet20.14 AmplexRed H2O2-production | 2019-06-24 | O2k-FluoRespirometry: HRR and simultaneous determination of H2O2 production with Amplex UltraRed. | Mouse | Heart |
| Komlodi 2018 Methods Mol Biol | 2018 | Komlodi T, Sobotka O, Krumschnabel G, Bezuidenhout N, Hiller E, Doerrier C, Gnaiger E (2018) Comparison of mitochondrial incubation media for measurement of respiration and hydrogen peroxide production. Methods Mol Biol 1782:137-55. | Human Mouse | Skeletal muscle HEK |
| Makrecka-Kuka 2015 Biomolecules | 2015 | Makrecka-Kuka M, Krumschnabel G, Gnaiger E (2015) High-resolution respirometry for simultaneous measurement of oxygen and hydrogen peroxide fluxes in permeabilized cells, tissue homogenate and isolated mitochondria. https://doi.org/10.3390/biom5031319 | Human Mouse | Heart Nervous system HEK |
| Krumschnabel 2015 Methods Mol Biol | 2015 | Krumschnabel G, Fontana-Ayoub M, Sumbalova Z, Heidler J, Gauper K, Fasching M, Gnaiger E (2015) Simultaneous high-resolution measurement of mitochondrial respiration and hydrogen peroxide production. Methods Mol Biol 1264:245-61. | Mouse | Nervous system |
MitoPedia concepts: MiP concept, SUIT protocol, Recommended
MitoPedia methods:
Fluorometry