Description
Abbreviation: Digitonin test
Reference: A: Optimization of digitonin concentration for pce
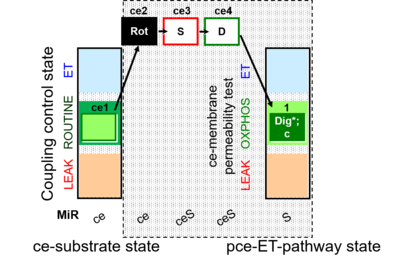
- SUIT protocol pattern: ce1;ce2Rot;ce3S;ce4D;1Dig;1c
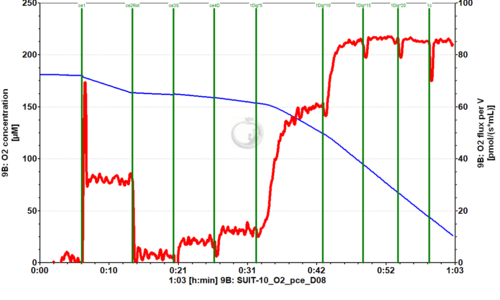
SUIT-010 is designed for the evaluation of optimum digitonin concentration for permeabilizing cells, a requirement to account for differences between cell types, the concentration of cells, and variability between batches of the natural product digitonin. After inhibition of the endogenous ROUTINE respiration by rotenone, respiration of living cells is not stimulated by succinate and ADP. Subsequent stepwise digitonin titration yields gradual permeabilization of the cell membrane, indicating by the increase of respiration up to full permeabilization. Respiration is constant over optimal digitonin concentration and inhibited by over-titration of digitonin, since the mitochondrial outer membrane is affected and cytochrome c can be released. Therefore, it is recommended to add cytochrome c after the optimal digitonin concentration had been reached. In other SUIT protocols for permeabilized cells, the respiration of pce is stable, if the optimal digitonin concentration is used.
Communicated by Doerrier C, Komlodi T, Cardoso LHD, Huete-Ortega M, Gnaiger E (last update 2019-06-05)
Specific SUIT protocols
SUIT-010 O2 ce-pce D008
- SUIT-010 O2 ce-pce D008 is a protocol to test the optimum concentration of digitonin
Steps and respiratory states
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| ce1 | ROUTINE | ce1
| ||
| ce2Rot | ROX | ce1;ce2Rot
| ||
| ce3S | SL(n) | S | CII | ce1;ce2Rot;ce3S
|
| ce4D | SP | S | CII | ce1;ce2Rot;ce3S;ce4D
|
| 1Dig | SP | S | CII | ce1;ce2Rot;ce3S;ce4D;1Dig
|
| 1c | SP | S | CII | ce1;ce2Rot;ce3S;ce4D;1Dig;1c
|
- Bioblast links: SUIT protocols - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- Coupling control
- Pathway control
- Main fuel substrates
- » Glutamate, G
- » Glycerophosphate, Gp
- » Malate, M
- » Octanoylcarnitine, Oct
- » Pyruvate, P
- » Succinate, S
- Main fuel substrates
- Glossary
Strengths and limitations
- Digitonin is a natural product and thus the effective concentration has to be determined by titrations for every batch. The optimum effective digitonin concentrations for complete plasma membrane permeabilization can be determined with this protocol.
- Plasma membrane permeabilization is faster than mitochondrial isolation.
- This protocol might be used simultaneously for selecting optimum experimental cell concentrations.
Compare SUIT protocols
- SUIT-003 have been designed to study the coupling control state of living cells
- SUIT-003 O2 ce D028 coupling-control protocol with succinate for permeability test
- SUIT-006 protocol is a coupling-control protocol for mitochondrial preparations
References
| Year | Reference | Organism | Tissue;cell | |
|---|---|---|---|---|
| Doerrier 2018 Methods Mol Biol | 2018 | Doerrier C, Garcia-Souza LF, Krumschnabel G, Wohlfarter Y, Mészáros AT, Gnaiger E (2018) High-Resolution FluoRespirometry and OXPHOS protocols for human cells, permeabilized fibers from small biopsies of muscle, and isolated mitochondria. Methods Mol Biol 1782:31-70. https://doi.org/10.1007/978-1-4939-7831-1_3 | Human Mouse Rat Saccharomyces cerevisiae | Heart Skeletal muscle Endothelial;epithelial;mesothelial cell Blood cells HEK Platelet |
MitoPedia concepts: MiP concept, SUIT protocol, Recommended
MitoPedia methods:
Respirometry